Age – Adult (18-75)
Gender – Female, Male
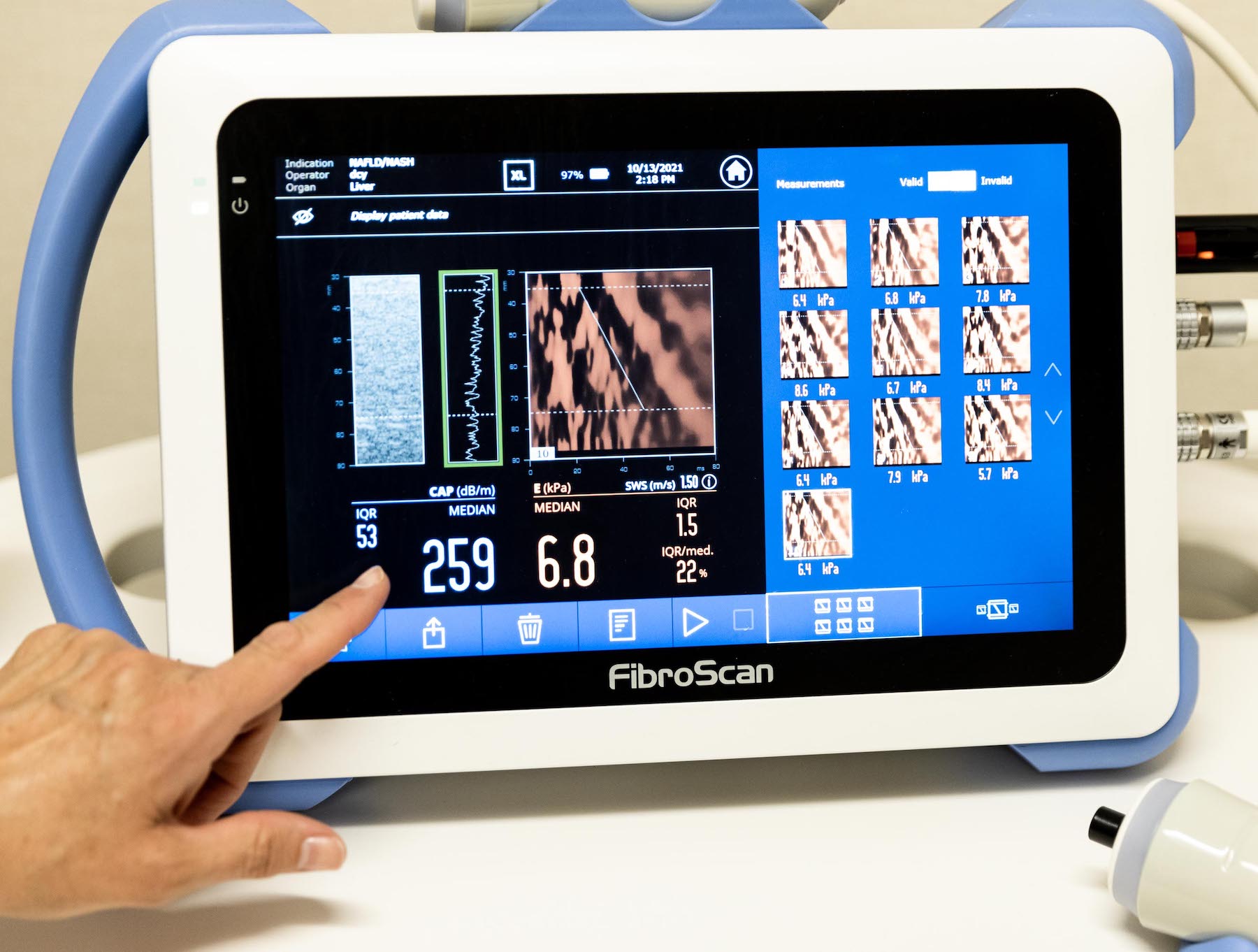
Disease Type – Primary Biliary Cholangitis
Phase – 3
Length – 52 weeks
Compensation available.
Inclusion:
– Male or female with a diagnosis of PBC based on history of any 2 of the following
criteria:
a. ALP above 1.0×ULN for at least 6 months
b. Positive AMA titer (>1:40 on immunofluorescence or M2 positive by ELISA) or
positive PBC-specific ANA
c. Documented liver biopsy results consistent with PBC
criteria:
a. ALP above 1.0×ULN for at least 6 months
b. Positive AMA titer (>1:40 on immunofluorescence or M2 positive by ELISA) or
positive PBC-specific ANA
c. Documented liver biopsy results consistent with PBC
– Non-pregnant females
Exclusion:
– Advanced PBC as defined by the Rotterdam criteria
– Other chronic liver diseases
– Known history of HIV
– History of drug abuse
– Cancer diagnosis
CymaBay: A 52-week, Double-blind, Placebo-controlled, Randomized, Phase 3 Study Intended to Determine the Effects of Seladelpar on Normalization of Alkaline Phosphatase Levels in Subjects with Primary Biliary Cholangitis (PBC) and an Incomplete Response or Intolerance to Ursodeoxycholic Acid (UDCA)
Learn More
Additional Information
For more information on what current clinical trials are available at Kansas City Research Institute or additional information and how to get involved, please contact our office phone at (816)759-2574 or send by email at study@kcri1.wpenginepowered.com.